LYME SCI: Do long COVID and long Lyme both activate mast cells?
Two new studies have shown a correlation between so-called “long COVID” and mast cell activation syndrome (MCAS).
This suggests that inexpensive, over-the-counter, mast cell stabilizing medications may be beneficial to people suffering from persistent effects of the coronavirus.
And it turns out, this same class of mast cell stabilizing medications may help Lyme patients as well.
In the first study, researchers found that patients with long COVID have many of the same symptoms as patients with mast cell activation syndrome (MCAS).
In the second, researchers gave COVID-19 patients MCAS medications early after infection and found far fewer developed severe illness (hospitalization) or long COVID.
The COVID-19 pandemic is roaring into its third year, with no end in sight. As data unfolds in real time, it appears that more than half of patients infected with SARS-CoV-2 go on to develop at least one chronic symptom lasting six or more months. (Logue et al, 2021) The medical name for this condition is post-acute sequelae of Covid-19 (PASC), also referred to as “long COVID.”
Similarly, the most recent research shows 43% of patients with prior Lyme disease have lingering symptoms following treatment (14% meeting the stricter research criteria of PTLDS,) a condition many people have started referring to as “long Lyme.” (Aucott, 2022)
Similarities
For COVID patients who survived ICU care, some 75% experience continuing symptoms after one year. (Hesakkers, 2022) Likewise for patients with delayed diagnosis for Lyme (disseminated Lyme). 73% rate their quality of life as fair or poor due to chronic symptoms. (Johnson et al, 2014)
Now, I’m not saying that Lyme and COVID are caused by the same pathogen. Far from it. Lyme is caused by spiral-shaped bacteria called Borrelia burgdorferi, while COVID is caused by the SARS-CoV-2 virus.
And in a perfect world, we would have targeted therapies—an antiviral for COVID, and an antibiotic for Lyme— able to eradicate both of these pathogens. Alas, those therapies remain evasive.
Many physicians with experience treating complex illness are finding it hard to ignore the symptoms common to both diseases—symptoms that may be relieved by calming the mast cells.
The MCAS hypothesis
Dr. Leonard Weinstock, a gastroenterologist at Missouri Baptist Medical Center, is a leading MCAS researcher. He and his colleagues hypothesize that the hyper-inflammation seen in COVID-19 may be caused by abnormally high mast cell activity.
In this study, researchers recruited 136 long COVID patients from a Facebook support group. In a survey, these patients were asked to compare their symptoms before and after having COVID-19.
Their survey responses were then compared to those of 80 patients known to have MCAS, and those of 136 healthy controls. The results were published in the International Journal of Infectious Diseases.
In the study, the researchers found a high prevalence of MCAS symptoms in long COVID patients prior to initiating treatment for mast cell activation.
According to Dr. Weinstock, before getting infected with COVID, patients in that group were very similar to the control group.
However, he said, “after the infection and when they developed long COVID, they had symptoms almost identical and similar in severity to those of mast cell activation patients.”
The article explains, “when major stressors drive escalations of MCAS, those escalations tend to be sustained for long/indefinite periods and sometimes clearly are permanent.”
What are mast cells?
Think of mast cells as universal alarm cells or guards. When mast cells detect infection, allergic triggers, injury, or toxins, they release specific chemicals (mediators) which trigger an immune response.
These mediators—which include histamine, cytokines, prostaglandin, tryptase, nitric oxide, heparin and many others—can cause a snowball effect that activates more mast cell reactions, triggering the release of more mediators.
Mast cells also express a substance called angiotensin converting enzyme 2 (ACE2), now known as the primary cell through which SARS-CoV-2 enters the respiratory tract (Theoharides, 2020).
[I have written a great deal about mast cell diagnosis and treatment after a diagnosis of MCAS helped solve the puzzle of my daughter’s prolonged illness caused by tick-borne disease.]
Symptoms of long COVID and long Lyme
There are no published surveys on the connection between MCAS and long Lyme. However, if you look at the top symptoms from the MyLymeData registry you’ll see a striking similarity to the symptoms of MCAS, Lyme and long COVID.
Does this mean that some of their common symptoms may be based on the same immune dysfunction—MCAS?
Let’s take the worst symptoms of Lyme as listed in MyLymeData. The most frequently reported symptoms of patients with chronic Lyme are fatigue, sleep impairment, joint/muscle pain, depression, cognitive impairment (brain fog), neuropathy, headaches, and heart-related problems . (Johnson 2019)
Compare that to the top symptoms of MCAS patients: fatigue, joint/muscle pain, dizziness/syncope, headache, hives/itchiness, numbness/tingling (neuropathy), nausea/vomiting, chills. (Afrin 2016)
And the symptoms of the long COVID patients: fatigue, loss of smell/taste, headaches, memory/cognitive impairment (brain fog), shortness of breath, increased heart rate or palpitations, muscle/joint pain, sleep impairment, nerve pain (neuropathy), sweats/chills, nausea or stomach pain. (Weinstock, 2021)
As you can see, all the top symptoms in long Lyme are present in both MCAS and long COVID.
Early signs of MCAS?
During her healing journey, we discovered my daughter had a predisposition to MCAS. Turns out she has an uncle with the condition. Looking back, the earliest signs I remember were her allergic reactions to the buttons on her infant pajamas and the elastic on her socks.
Knowing how diagnosis and treatment of mast cell activation helped my daughter, I believe this information bears repeating. Had we known what we know today, her doctor and I both would have started MCAS treatment prior to beginning antibiotic therapy. We both feel that would have lessened the extreme Herxheimer reactions.
Of course, the cause of chronic illness varies from person to person. For example, we know that with Lyme disease, patients who are co-infected with another pathogen (eg. Babesia, Bartonella, or even a reactivated virus), are much sicker than those dealing with a single pathogen.
Pre-existing conditions
The same is true for COVID. Those living with a pre-existing condition (eg. diabetes, high blood pressure, obesity), and those dealing with a co-infection or reactivated virus, like Epstein Barr, are at much higher risk of worse outcomes from COVID.
At least in some, the root cause of long COVID and long Lyme may be due to a preexisting or genetic predisposition to MCAS. The additional stress of infection may also lead to mutations in stem cells and abnormal mast cells. It’s estimated that 10-30% of the U.S. population has MCAS.
It is also reasonable to say that a great many cases of MCAS are rooted in an ongoing (acute, chronic or stealth) infection.
Weinstock and his team believe that long COVID symptoms could be mitigated by preventing or calming the mast cell activation at the onset of illness by using drugs commonly used to treat MCAS.
“This brings up the possibility that what we use to treat Mast Cell Activation Syndrome patients may also help long COVID patients,” said Dr. Weinstock.
Testing out the MCAS theory
In the second study, researchers set out to test whether two common MCAS medications could reduce the severity of COVID and prevent many aspects of long COVID. Their work was published in the Journal of International Immunopharmacology.
In this pilot study, researchers enrolled 53 COVID patients from a single center in Boston during the first wave of COVID in 2020. The patients were given a combination of two over-the-counter MCAS medications—levocetirizine (Brand name Xyzal) and montelukast (Brand name Singulair)—and followed for six months. (May, Gallivan, 2022)
The authors report, “Fifty-one of 53 patients were considered a clinical cure on therapy with restoration of their overall status to a pre-infection baseline within 2 weeks. Two patients, ages 73 and 80, continued to complain of fatigue for a period of time post discontinuation of therapy.”
Among the patients enrolled in this study, none required intubation and there were no deaths. When compared to the general population at the time with a 1%-7.5% mortality rate, this appears to be an improvement.
Treating MCAS
Several allergy and indigestion medications are commonly used to treat MCAS. In the first study, the authors reported seeing improvement in their long COVID patients with medications used to treat MCAS including:
- Antihistamines
- Cromolyn
- Flavonoids (quercetin and lutein)
- Low dose naltrexone
- Montelukast, and
- Vitamins C and D
“Low dose naltrexone has also been used to treat MCAS and may be effective in long COVID, possibly by reducing cytokines from T-cells which activate mast cells and by blocking Toll-like receptors on mast cells and microglia,” the report states.
In addition, to the above trials, I’d like to point out a prior study by Jayakumar Rajadas, PhD, of the Stanford School of Medicine. It found that a common antihistamine used in MCAS treatment was effective in starving and killing Borrelia burgdorferi in the laboratory.
The results of Dr. Rajadas’s study showed that Loratadine (brand name: Claritin) could prevent manganese from entering the cell wall of Borrelia in the laboratory. Unlike most bacteria, Borrelia uses manganese to evade the immune system, rather than iron. Loratadine blocks the manganese by inhibiting the bacteria’s transport system, BmtA (Borrelia metal transporter A). The study was published in the open access publication Drug Design, Development and Therapy.
A more recent study by Dr. David Ostrov, an immunologist and associate professor in the University of Florida College of Medicine’s, has shown great promise in COVID treatment by combining two over-the-counter medications.
Dr. Ostrov’s study, which combined the antihistamine diphenhydramine (common name: Benedryl), and lactoferrin, a protein in milk, showed 99% efficacy in inhibiting replication of the SARS-CoV-2 virus. His research was published in the journal Pathogens.
“My prediction is that antiviral drug combinations, such as diphenhydramine and lactoferrin, will provide a similar level of benefit as Regeneron monoclonal antibodies, Pfizer and Merck antivirals, at less than 1/100 the cost of those therapies,” says Dr. Ostrov.
(See my previous blog suggesting that Lyme disease patients may also benefit from mast cell stabilizers early in their treatment.)
Mast cells in the brain
Mast cells are found on the brain side of blood vessels, allowing them to communicate with neuroglia. Neuroglia are highly branched cells that occupy the spaces between neurons and comprise all the non-nerve cells of the brain and spinal cord.
Four main types of neuroglia are recognized: astrocytes, microglia, oligodendrocytes, and ependymal cells. Increased glial activity and neuroinflammation in the brain lead to brain dysfunction.
Low dose naltrexone (mentioned above) is also a glial inhibitor, as-is minocycline an antibiotic frequently used in the treatment Lyme and other tick-borne diseases.
Mast cells and microglial inflammation contribute to the exacerbation of acute symptoms of Lyme disease and COVID and can accelerate neurodegenerative disease as well as promoting the pain pathway via the vagus nerve.
Inhibiting mast cells and microglial inflammation in the brain may be key to reducing the cognitive and psychological symptoms of long Lyme and long COVID.
Hope for the future
The researchers believe this first step may lead to more effective ways to treat long COVID and long Lyme patients.
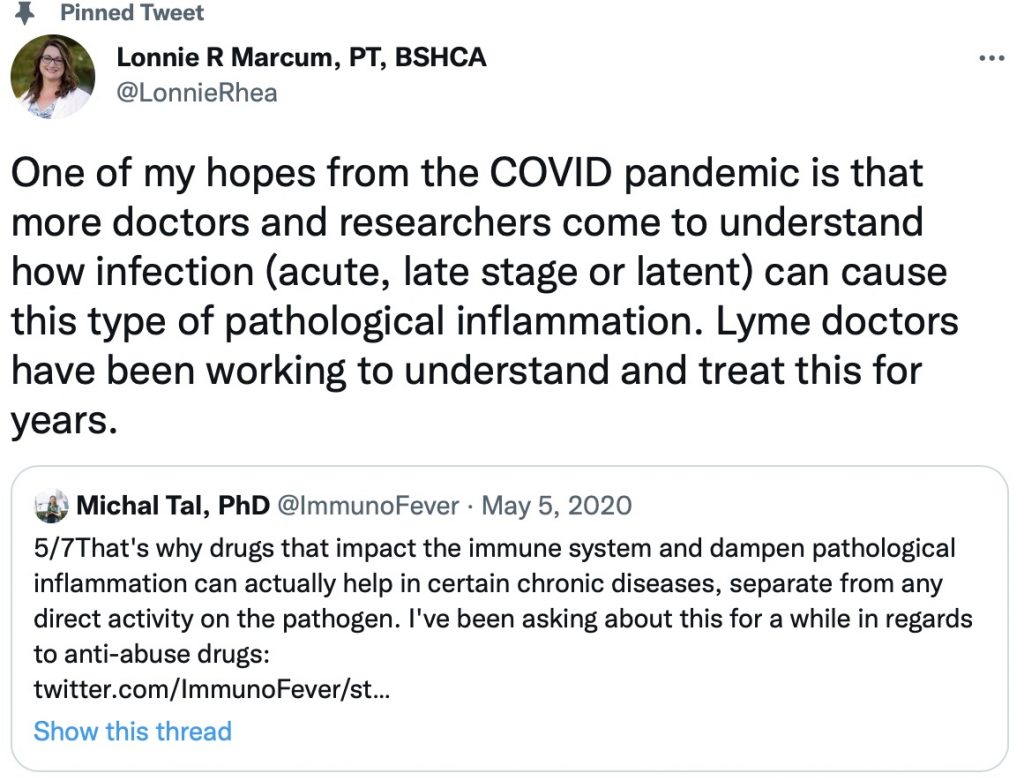
In May 2020, I tweeted in response to researcher Dr. Michal Tal: “One of my hopes from the COVID pandemic is that more doctors and researchers come to understand how infection (acute, late stage or latent) can cause this type of pathological inflammation. Lyme doctors have been working to understand and treat this for years.”
What this research tells us is that many medications already FDA-approved may be of benefit when administered early in the disease process. If you have prolonged mast cell symptoms following an acute infection, you may want to explore a diagnosis and treatment for MCAS with your practitioner.
LymeSci is written by Lonnie Marcum, a Licensed Physical Therapist and mother of a daughter with Lyme. She serves on a subcommittee of the federal Tick-Borne Disease Working Group. Follow her on Twitter: @LonnieRhea Email her at: lmarcum@lymedisease.org.
References
Afrin, L.B., Weinstock, L.B., Molderings, G.J. (2020) Covid-19 hyperinflammation and post-Covid-19 illness may be rooted in mast cell activation syndrome. Int’l J of Infectious Diseases. Volume 100, Pages 327-332. DOI https://doi.org/10.1016/j.ijid.2020.09.016
Afrin LB, Self S, Menk J, Lazarchick J. (2016) Characterization of Mast Cell Activation Syndrome. Am J Med Sci. 2017 Mar;353(3):207-215. doi: 10.1016/j.amjms.2016.12.013.
Aucott JN, Yang, T, Yoon I, Powell D, Geller SA, Rebman AW (2022) Risk of post-treatment Lyme disease in patients with
ideally-treated early Lyme disease: A prospective cohort study, Int’l J. of Infectious Diseases (2022), doi: https://doi.org/10.1016/j.ijid.2022.01.033
Erol AYG (2015) The Role of Mast Cells and Neuroglia in Neuroinfectious Diseases. J Neuroinfect Dis 6:190. doi:10.4172/2314-7326.1000190
Heesakkers H, van der Hoeven JG, Corsten S, et al. Clinical Outcomes Among Patients With 1-Year Survival Following Intensive Care Unit Treatment for COVID-19. JAMA. Published online January 24, 2022. doi:10.1001/jama.2022.0040
Johnson L, Wilcox S, Mankoff J, Stricker RB. (2104) Severity of chronic Lyme disease compared to other chronic conditions: a quality of life survey. PeerJ. 2014; 2. Available from: https://dx.doi.org/10.7717/peerj.322.
Johnson, L. (2019): 2019 Chart Book — MyLymeData Registry. (Phase 1 April 27, 2017. Sample 3,903). figshare. DOI: https://doi.org/10.6084/m9.figshare.7849244
Logue JK, Franko NM, McCulloch DJ, et al. Sequelae in Adults at 6 Months After COVID-19 Infection. JAMA Netw Open. 2021;4(2):e210830. doi:10.1001/jamanetworkopen.2021.0830
May BC, Gallivan KH. Levocetirizine and montelukast in the COVID-19 treatment paradigm. Int Immunopharmacol. 2022 Feb;103:108412. doi: 10.1016/j.intimp.2021.108412. Epub 2021 Dec 15. PMID: 34942461; PMCID: PMC8673734.
Ostrov, DA., et al. (2021) “Highly Specific Sigma Receptor Ligands Exhibit Anti-Viral Properties in SARS-CoV-2 Infected Cells” Pathogens 11:1514. DOI: https://doi.org/10.3390/pathogens10111514
Skaper, S. D., Facci, L., & Giusti, P. (2014). Mast cells, glia and neuroinflammation: partners in crime?. Immunology, 141(3), 314–327. https://doi.org/10.1111/imm.12170
Theoharides T. C. (2020). COVID-19, pulmonary mast cells, cytokine storms, and beneficial actions of luteolin. BioFactors (Oxford, England), 46(3), 306–308. https://doi.org/10.1002/biof.1633
Wagh D, Pothineni V, Inayathullah M, Liu S, Kim K, Rajadas J. (2015) Borreliacidal activity of Borrelia metal transporter A (BmtA) binding small molecules by manganese transport inhibition. Drug Des Devel Ther. 2015;9:805-816 DOI: https://doi.org/10.2147/DDDT.S77063
Weinstock, LB, Brook, JB, et. al. (2021) Mast cell activation symptoms are prevalent in Long-COVID. Int’l J of Infectious Dis. 112(2021), 217-226, DOI: https://doi.org/10.1016/j.ijid.2021.09.043.





















We invite you to comment on our Facebook page.
Visit LymeDisease.org Facebook Page