T he blacklegged tick—Ixodes scapularis on the East Coast and Ixodes pacificus in the West—is the primary vector for Lyme disease in the United States. It can also transmit several other pathogens, commonly known as co-infections.
One of those co-infections is called anaplasmosis, which, according to a large survey conducted by LymeDisease.org, occurs in about 5% of patients with Lyme disease. (see my previous post on co-infections here.)
Human Granulocytic Anaplasmosis
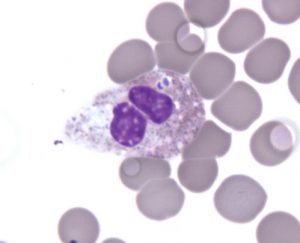
Anaplasma phagocytophilum is commonly found in rodents and small mammals, and can be transmitted to humans by the bite of an infected tick.
The number of reported cases of HGA has been steadily increasing in recent years, with 2800 confirmed cases in 2014. However, these numbers are likely to be low for several reasons. Many cases go undiagnosed or are not confirmed with laboratory testing, and not all states require physicians to officially report the illness.
Anaplasmosis Geographic Distribution
The geographic distribution for HGA is very similar to Lyme, because both diseases share the same primary hosts and primary vectors. The six states with the highest incidence for HGA are Rhode Island, Minnesota, Connecticut, Wisconsin, New York, and Maryland.
The incidence of anaplasmosis is largely underreported, based on several recent studies.
Mr. Ostfeld feels that the incidence of anaplasmosis is largely underreported, based on several recent studies. In one two-year study, 45% of wild white-footed mice tested positive for Lyme disease, Babesia, and Anaplasma, suggesting high exposure rates to ticks that feed on them. Another study suggested that Anaplasma may act as a type of anti-freeze that helps ticks survive freezing temperatures.
Overall, the infection rate of Anaplasma within questing nymphal ticks varies greatly by state, but has been detected from coast to coast.
West Coast: 0.8%–4.2%
East Coast: -5%–8%
Signs and Symptoms
The symptoms of HGA usually appear 5-14 days after the tick bite and typically include the following:
- Fever (92-100%)
- Headache (82%)
- Fatigue (97%)
- Muscle pain (77%)
- Shaking/chills (77%)
- Rash (fewer than 10%)
- Gastro-intestinal symptoms (rare)
- Neurological symptoms (rare)
Clinical Course
The majority of HGA cases are mild and self-limiting. Infrequently, however, complications like respiratory failure, peripheral neuropathies, bleeding/coagulation problems, pancreatitis, and kidney failure may occur. Rarely, HGA can be fatal.
A delay in treatment can result in severe symptoms that may initially resemble toxic shock syndrome—a complication that sometimes arises from certain bacterial infections.
Other serious viral or fungal infections can occur during the course of infection while the immune system is compromised. Some HGA patients may need to be hospitalized.
Co-infection with other diseases transmitted by Ixodes ticks (Lyme disease, Borrelia miyamotoi, Babesia, or Powassan virus) can complicate the course of HGA. If a patient has a delayed response to Lyme treatment and a low white blood cell count, it should raise suspicion for possible co-infection with Anaplasma.
Anaplasmosis Treatment

In addition, “If the patient is treated within the first five days of the disease, fever generally subsides within 24-72 hours… Severely ill patients may require longer periods before their fever resolves.”
Summary of HGA
Lonnie Marcum is a licensed physical therapist and mother of a daughter with Lyme. Follow her on Twitter: @LonnieRhea. Email her at lmarcum@lymedisease.org.
References:
- Forbidding Forecast for Lyme Disease in the Northeast
- CDC MMWR | Diagnosis and Management of Tickborne Rickettsial Diseases: Rocky Mountain Spotted Fever and Other Spotted Fever Group Rickettsioses, Ehrlichioses, and Anaplasmosis — United States
- CDC | Emerging Tickborne Diseases | Public Health Grand Rounds, March 2017
- Serum Antibodies to Borrelia Burgdorferi, Anaplasma Phagocytophilum, and Babesia Microti in Recaptured White-Footed Mice
- Co-Infection of Blacklegged Ticks with Babesia Microti and Borrelia Burgdorferi is Higher than Expected and Acquired from Small Mammal Hosts
- Epidemiology and Genetic Diversity of Anaplasma Phagocytophilum in the San Francisco Bay Area, California
- Anaplasma Phagocytophilum Induces Ixodes Scapularis Ticks to Express an Antifreeze Glycoprotein Gene that Enhances their Survival in the Cold.




















