LYMEPOLICYWONK: Patients can’t wait, whether it’s COVID-19 or Lyme

Even a few short weeks ago, who would have thought we’d be wearing face masks and gloves to visit a grocery store? Or that any shopper would sport a get-up like the one in this picture?
COVID-19 is changing the world as we know it. This coronavirus will change the world for patients with chronic Lyme disease, too.
U.N. Secretary-General Antonio Guterres now warns that with COVID-19, the world faces its most challenging crisis since World War II in terms of human suffering, deaths, and economic disruption.
Patients need treatments and they need them now. The grindingly slow pace of traditional research won’t do. We need guerrilla research that is fast, agile and “good enough” for now. Research innovation is long overdue. It will benefit all patients everywhere, including those who have Lyme disease.
In a recent Forbes interview about COVID-19, I said “It is important right now to take the gloves off clinicians and give them access to all available tools. Patients are dying and can’t wait for clinical trials.”
My comment to Forbes about the need to act quickly drew some heat. Some people countered that we need evidence BEFORE we can treat and that physician reports of what treatments they see working in hospitals are mere anecdotes.
Sound familiar? Since 2006, that’s what the Infectious Disease Society of America has been saying in its guidelines to deny treatment to Lyme patients who remain ill. The COVID-19 pandemics shines a spotlight on the need to act in the face of uncertainty. Same issue, different disease.
Evidence-based medicine, COVID-19 and chronic Lyme disease
Evidence-based medicine is the combination of scientific evidence, clinical judgement, and patient preferences. Researchers tend to focus on the evidence piece of the puzzle. They insist that treatment be validated in randomized controlled trials before anyone can use it.
That’s fine for researchers who aren’t sick with the disease. But for patients and the doctors who treat them, the question is: what treatments are worth trying? Specifically, are the risks of any given treatment worth it, given the potential benefits? The answer to that depends in part on how sick the patient is and what their treatment alternatives are.
For patients without treatment options, like those with COVID-19 and persistent Lyme disease, the amount of risk that they are willing to take on typically depends upon the severity of their illness or the degree to which their quality of life is impaired.
Real-world data collected at the bedside by clinicians, their institutions, or patients provides valuable information. Yet, it can’t demonstrate cause and effect. It can’t tell us whether a patient got well because of a particular treatment. This patient might have had a milder form of the disease, had a better immune system, or the doctor might have only given the treatment to certain patients.
To demonstrate cause and effect, randomized controlled trials (RCTs) are required. The problem with RCTs, Dr. Rob Califf, former head of the FDA explains, is that “they take too long, cost too much, and don’t apply to most people.” In a crisis, barriers like these cost lives.
Anecdotes vs evidence: the hydroxychloroquine controversy
The issue of how much evidence is enough for a doctor to act takes on new urgency with COVID-19’s mounting death toll. It has erupted in full force over the innovative use of hydroxychloroquine (HCQ) (also known as Plaquenil), sometimes in combination with azithromycin (H&Z). (Doctors sometimes use these drugs to treat persistent Lyme disease, although not necessarily in combination.)
The tone of the debate around H&Z is heightened by its political overtones. President Trump has encouraged people to use the treatment, asking rhetorically “what can it hurt?” Meanwhile, others have derided H&Z as “scientifically unproven.” They note that (like all medical treatments) there may be side-effects, some serious.
LymeDisease.org is not weighing in on this debate one way or the other. Instead, we look at the healthcare policy arguments advanced generally to see how they apply to persistent Lyme disease.
COVID-19 highlights the tension between doctors in the trenches battling illness and researchers sitting in ivory towers focusing on abstract concepts that don’t land in the real world with patients whose lives are at risk.
The clinical experience of doctors is an essential form of scientific knowledge that is being dismissed by researchers who don’t bear the consequences of failing to intervene when people are sick. These researchers don’t have to tell the families who have suffered a loss that they didn’t do everything they could to help.
We see the divide between academic researchers and physicians who treat in Lyme disease every day, but the story has not resonated with the media or the public in the way the COVID-19 crisis does.
COVID-19 underscores the importance of clinical innovation to save lives. New therapies may take a year or so to develop. In contrast, doctors can repurpose FDA-approved drugs off-label for use with COVID-19 today. Some doctors have reported success treating COVID-19 with HCQ or H&Z—reports which some researchers dismiss as mere “anecdotes.” However, a survey of over 6,000 doctors found that azithromycin (41%) and hydroxychloroquine (33%) are among the three most commonly prescribed treatments for COVID-19.
False dichotomy
In a recent article, Dr. Perry Wilson, Associate Professor at Yale School of Medicine, called out the false dichotomy of framing the HCQ debate as “evidence vs anecdote”:
“The bottom line is that we don’t need to abandon evidence-based medicine in the face of the pandemic. We need to embrace it…and… realize: Evidence-based medicine is not just about randomized trials; it’s about appreciating the strengths and weaknesses of all data, and allowing the data to inch us closer and closer toward truth. …We don’t have the luxury of waiting for randomized controlled trials. We need to act on the data we have. As more data come in, we can revise those estimates of efficacy, iteratively and transparently.”
Wilson’s piece garnered over 129 comments about whether doctors should provide treatment that “might work” before the evidence is settled.
One clinician put the need for doctors to act in stark terms: “This is war. In war, we do not wait for a RCT to see if a particular new weapon will work. If it works and kills the enemy – good. If it does not work, bad luck. If it works adversely, bad luck – the soldiers were going to die of enemy fire anyway. In [this] case the new weapon is in fact, an old weapon established as effective against another type of enemy, so no harm in trying it.” This is the approach many Lyme doctors take.
These arguments should sound familiar. Lyme patients and their treating clinicians have been saying this for years. We make these arguments in the context of Lyme disease in my subcommittee’s report to the federal Tick-Borne Disease Working Group (TBDWG). This spells out the need to make decisions in Lyme disease even when science is uncertain.
Shared medical decision-making
Putting aside the political theatrics in the H&Z controversy, the full spectrum of the debate on what to do when science is uncertain is represented on the President’s own COVID-19 team. Trump’s encouragement of the use of H&Z has been branded as anti-science. At the same podium, his advisor Dr. Anthony Fauci (the director of the National Institute of Allergy and Infectious Diseases), calls the treatment unproven. Dr. Deborah Birx, (the Coronavirus Response Coordinator) says that the decision does not belong to either President Trump or Fauci, but rather belongs to the individual patient and their doctor. This is known as shared medical decision-making.
We see this same range of views in the treatment of persistent Lyme disease, with ILADS physicians being accused of using “unproven treatments” and the IDSA asking patients to wait until the science is in. But the Lyme disease treatment research boat has not set sail for over 20 years, when the last clinical trial for treating persistent Lyme disease was funded.
Unlike COVID-19, Lyme disease is a research-disadvantaged disease—with very little government or industry interest in developing new treatments. If COVID-19 research is a fire hose full blast right now, then Lyme disease research is a parched desert. The sad fact is that without incentives for research, Lyme disease is unlikely to achieve scientific certainty any time soon and patients who are sick today need treatment.
In his article, Wilson says that where the evidence is not yet in and the patient outcome is uncertain, the “key to evidence-based medicine during this epidemic is being transparent about what we know and what we don’t. If we want to use hydroxychloroquine, that is a reasonable choice, but we need to tell the public the truth: We’re not too sure it will work, and it may even be harmful.”
That sounds like the calls in the Lyme community for shared-medical decision-making with patients. This is one of the recommendations contained in my subcommittee’s report to the TBDWG. The Working Group adopted it and will submit it to Congress as a call to action.
Recommendation 7.1: Recommend Federal government websites and educational materials and seminars for clinicians, the public, and public health departments, which discuss Lyme disease, provide information that the state of the science relating to persistent symptoms associated with Lyme disease, is limited, emerging, and unsettled; and increase public awareness that there are divergent views on diagnosis and treatment. Consider that shared medical decision-making may be appropriate in some circumstances.
Lyme disease has always played David to the IDSA’s Goliath in terms of power. The urgency of COVID-19 has brought the debate about the need to act when patients are ill and science is unsettled and the importance of shared medical decision-making into the mainstream. Shining the COVID-19 spotlight on these issues may help Lyme disease patients obtain the treatment they need.
The COVID-19 research renaissance
The COVID-19 crisis is transforming the speed and way that research is conducted. One physician commented to the article by Wilson saying, “this pandemic can be a real eye-opener. . .[on] the need to revolutionize regulatory infrastructure and come up with innovative solutions for the benefit of our patients.” This is a call for research to enter the 21st century and begin using big data tools, including the use of research registries.
Clinician and institutional registries are being used to capture data about COVID-19 cases. For example, a preprint of a study of over 4,000 patients at one New York center from March 1 to April 2 was published within just seven days! That study analyzed patients who were diagnosed, admitted, or became critically ill during that month.
This is why LymeDisease.org launched the MyLymeData registry, which has enrolled over 13,000 patients. Patient have long recognized the need to accelerate research and to recognize all forms of data as useful. Data from the registry has helped determine the ground truth for Lyme patients. (You can read results from the registry here and here.) You can enroll in the registry here.)
There has also been a virtual explosion in the number of research trials being conducted for COVID-19. More than 160 COVID-19 treatments and vaccines are either under development or consideration.
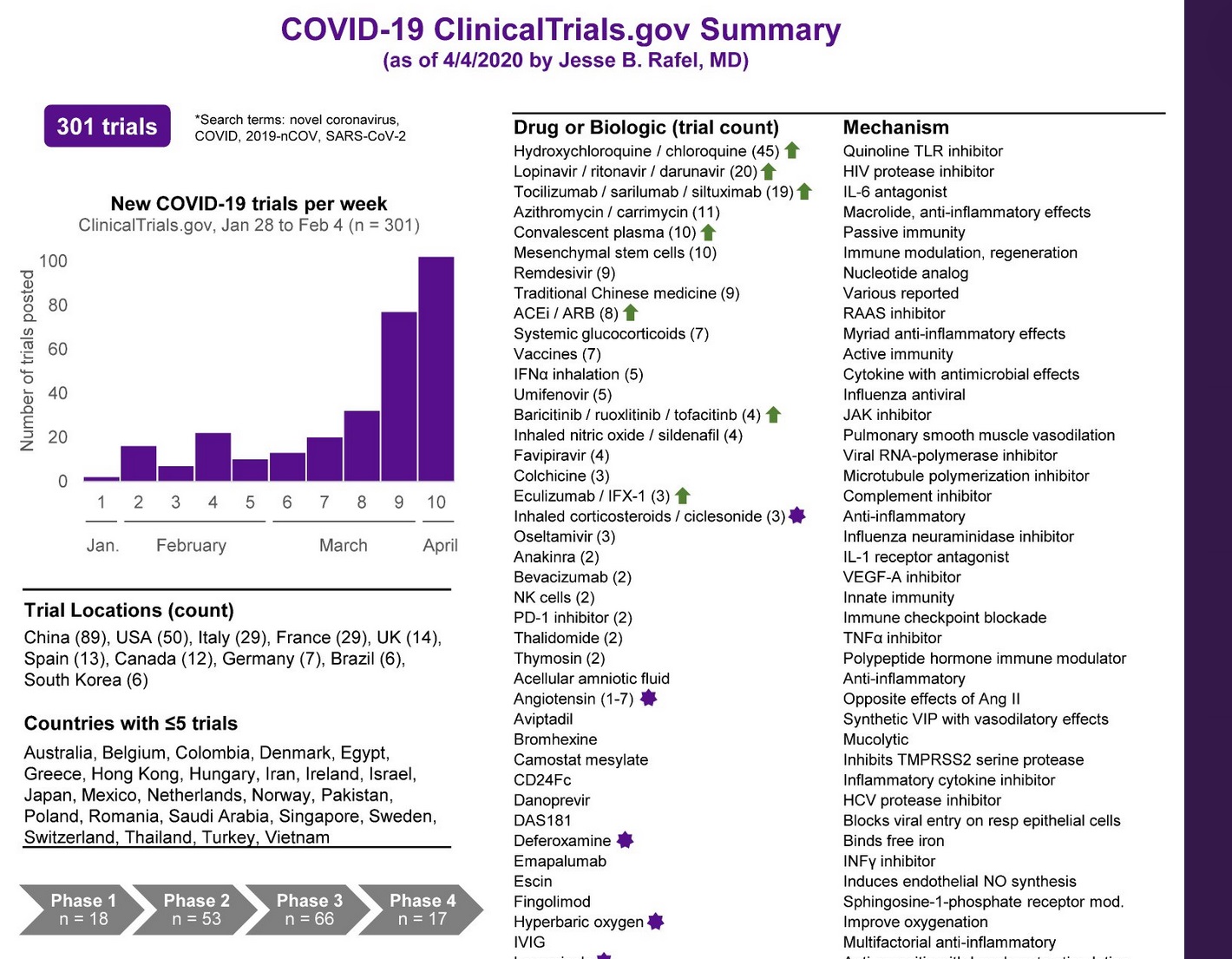
These include antivirals, antibiotics, anti-inflammatories, antibody-based treatments, and vaccines. They include drugs currently being developed as well as FDA-approved drugs that might be repurposed for treating COVID-19. And, 300 clinical trials are listed on ClinicalTrials.gov as of April 4, with more listed every week.
COVID-19 is accelerating changes in the way trials are conducted and in the use of innovative research tools, like big data. For example, Yale, Rush University in Chicago, and the University of Washington have proposed a trial called INSPIRE that would use real-world evidence in a national registry. The study is unique because it would allow data to be entered about treatment outcomes by many different stakeholders, including patients, emergency rooms, institutions, clinicians, and lab test sites.
This proposed study was presented at the NIH Collaboratory on April 3 (which focused on creating a rapid cycle learning research system where research is conducted quickly and implemented into clinical practice).
The importance of patient participation was captured in the slide below:
Another study presented on April 17 at the NIH Collaboratory called HERO seeks to enroll over 15,000 healthcare workers (who are at high risk of contracting COVID-19) to track their symptoms and challenges dealing with COVID-19.
It will also use 40 clinical research networks—institutions like Kaiser, for example—to see if using HCQ in healthcare workers prevents them from contracting the illness.
A recent commentary in the New England Journal of Medicine correctly points out that “it is a false dichotomy to suggest that we must choose between rapid deployment of treatments and adequate scientific scrutiny.”
COVID-19 is showing us that we do not need to settle on a single evidence-building approach. Instead, we can and must use all the tools at our disposal to solve this problem at the same time. These tools include:
- encouraging frontline clinical innovation on a patient to patient basis,
- using registries to capture results of innovative clinical approaches where possible, and
- conducting multi-site clinical trials, including RCTs, to evaluate treatment outcomes.
COVID-19 has ushered in a brave new world of research and heightened debates around the role of the doctor and patient in making health care decisions when evidence is uncertain. As COVID-19 is showing us, the full range of evidence encompasses patient experience, clinical experience, registry-gathered data of outcomes, and randomized controlled trials all exist along a continuum.
Valuing one does not mean devaluing the other forms of evidence. This type of innovation is a good model for Lyme disease. When the IDSA says “let’s wait for the evidence,” patients need to push back. Because COVID-19 patients can’t wait, and neither can patients with Lyme disease.
Lorraine Johnson, JD, MBA, is the Chief Executive Officer of LymeDisease.org. You can contact her at lbjohnson@lymedisease.org. On Twitter, follow her @lymepolicywonk.
References
Mary Beth Pfeiffer, This Coronavirus Patient Dodged A Bullet With Hydroxychloroquine. Is She A Harbinger Or Outlier? Forbes, March 22, 2020. https://www.forbes.com/sites/marybethpfeiffer/2020/03/22/one-patient-dodges-a-covid-bullet-is-she-a-harbinger-or-outlier/#4c9f7b975b84
Sermo, COVID-19 Real Time Barometer Study, WAVE I: March 25 -27, 2020. https://public-cdn.sermo.com/covid19/c8/be4e/4edbd4/dbd4ba4ac5a3b3d9a479f99cc5/wave-i-sermo-covid-19-global-analysis-final.pdf
- Perry Wilson, MD, MSCE, Hydroxychloroquine for COVID-19: What’s the Evidence? Medscape, March 25, 2020 https://www.medscape.com/viewarticle/927342
Alex Philippidis,Vanquishing the Virus: 160+ COVID-19 Drug and Vaccine Candidates in Development, Genetic Engineering and Biotechnology News, April 13, 2020, https://www.genengnews.com/a-lists/vanquishing-the-virus-160-covid-19-drug-and-vaccine-candidates-in-development/#Front_Runners
Rafel, Jesse. COVID-19 Clinicaltrials.gov Summary as of April 4, 2020. https://twitter.com/jbrafel/status/1247058257341972482/photo/1
NIH Collaboratory, April 3, 2020: Innovative Support for Patients with SARS-COV-2 Infections Registry (INSPIRE): Participant-Centered, Rapidly-Deployed, Digitally-Enabled Research (Harlan Krumholz, MD; Bala Hota, MD, MPH; Graham Nichol, MD, MPH). https://rethinkingclinicaltrials.org/news/april-3-2020-innovative-support-for-patients-with-sars-cov-2-infections-registry-inspire-participant-centered-rapidly-deployed-digitally-enabled-research-harlan-krumholz-md-bala-hota-md-mp/
NIH Collaboratory, April 17, 2020: The HERO Program: PCORnet® at Work to Create a Healthcare Worker Community for Rapid Cycle Evidence (Nakela Cook, MD, MPH; Josie Briggs, MD; Susanna Naggie, MD; Emily O’Brien, PhD; Russell Rothman, MD; Chris Forrest, MD, PhD). https://rethinkingclinicaltrials.org/news/april-17-2020-the-hero-program-pcornet-at-work-to-create-a-healthcare-worker-community-for-rapid-cycle-evidence-nakela-cook-md-mph-josie-briggs-md-susanna-naggie-md-emily-obr/
Benjamin N. Rome, M.D., and Jerry Avorn, M.D., Drug Evaluation during the Covid-19 Pandemic, NEJM, April 14, 2020. https://www.nejm.org/doi/full/10.1056/NEJMp2009457





















We invite you to comment on our Facebook page.
Visit LymeDisease.org Facebook Page